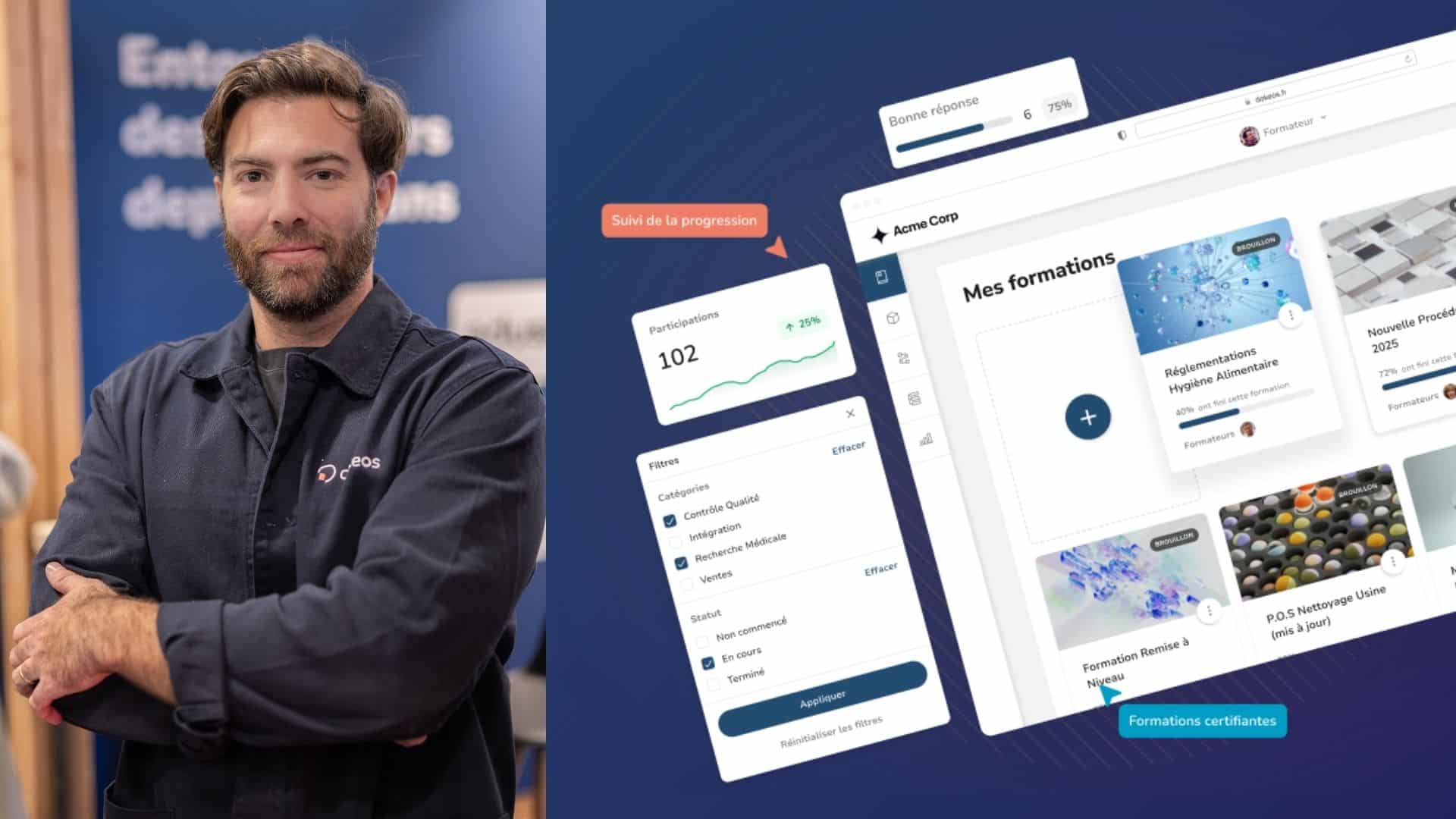
Many clinical and pharmaceutical research laboratories are training their staff using elearning. Discover the benefits and solutions of this approach.
To maintain an optimum level of quality and ensure their employees have the latest training, clinical and pharmaceutical research laboratories are increasingly calling on elearning. DOKEOS tools in particular allow for precise and intelligent employee evaluations.
Elearning for clinical and pharmaceutical research
Many companies and pharmaceutical businesses engaged in clinical research today rely on elearning, particularly with tools from the DOKEOS suite. When it comes to training employees, elearning offers many practical advantages.
Elearning and the quality approach
A qualitative approach is absolutely essential for clinical pharmaceutical research specialists. Elearning enables the development of comprehensive training courses, including the possibility for certification. It helps to ensure greater standardization of procedures, not to mention compliance. Regular training can ensure that each year, personnel are suitably upgraded and retrained.
The Quality Assurance department has a decisive role in this process. Usually this department is responsible for funding the training, but it also initiates the training protocols and evaluation that guarantee the company meets the knowledge and practices set by various national and international standards bodies: FDA 21 CFR part 11, GAMP5, ISO 9001, ASAE 3402.
Training for sales representatives
Sales rep jobs require regular training in sales and negotiation techniques as well as about the products being distributed. Mobile and often geographically dispersed, sales staff are not typically readily available for face-to-face training.
Elearning is a way to ensure consistency in training for sales representatives and help them to continually maintain their skills, all for a modest cost in terms of time and money. Elearning also allows for increased reactivity, particularly for example when new products are released.
Support for personnel
Companies and organizations involved in clinical research just as often turn to DOKEOS to implement other types of specific programs.
These might include, for example, analyzing and improving professional practices through the statistical measurement of prescription practices.
Elearning is also a good way to support new starters in the business through the provision of a standardized induction course that explains best practices, regulations, the procedures in place and contractual commitments.
DOKEOS EVALUATION, a powerful tool for clinical research and pharmaceuticals
Diverse training strategies
The tools provided by DOKEOS EVALUATION allow clinical and pharmaceutical research specialists to develop diverse types of questionnaires and evaluation forms:
- Self-training quizzes
- Certification examinations
- Questionnaires about practices, with statistical evaluations of results
- Surveys on the use of a drug by a given population
- Satisfaction surveys
- Feedback on professional practices for physicians and pharmacists
A tool for clinical research
In addition, questionnaires can be structured to take account of the particularities of clinical reasoning. For certain partners, DOKEOS has developed specific questionnaires, for example:
- Choice of medication substances in a list
- Conditional questions: for example, “If yes, how many grams?”
- Relative questions: for example, “If yes, proceed to question 3.”
Large companies invest in elearning
Clinical and pharmaceutical research laboratories including Alcon, Ipsen, Daiichi, Prostrakan and Thrombogenics all use elearning for employee training and evaluation processes, each relying on the power, reliability and flexibility of the DOKEOS ELEARNING SUITE.
You too can discover the exclusive features of DOKEOS during a free, 60-day trial.