The pharmaceutical sector is an ever-evolving industry, utilizing the latest in AI and machine learning to produce breakthroughs in medical science. While technology has been largely a force for good, it does require workers to keep up to date with the constant evolution in technology and regulations. The optimal solution is a remote learning environment that minimally disrupts daily work operations. Learn how Dokeos learning management system (LMS) can assist in the growth of pharma workforce skills and be a driver for innovation.
Why pharma workforce skills matter
According to a 2020 McKinsey report, technological disruption is the biggest inhibitor of employee growth and productivity in the pharma industry. Additional related factors like the transition to a direct-to-customer sales model have also caused disruptions. Simply put, technology and a change in environment are occurring at a greater pace than employers and workers can keep up. It’s estimated that an estimated 900,000 pharmaceutical jobs could disappear in the next 10 years due to automation. However, an estimated 90,000 to 120,000 new jobs will also be created in the process.
What’s the solution? The McKinsey report cited employee reskilling as the pathway for addressing the increasingly widening talent gap, particularly as it relates to technology, machine learning, and automation tools. Existing and incoming employees need continuing education to keep up with the changes in real time.
How can Dokeos LMS be a driving force for transformative learning?
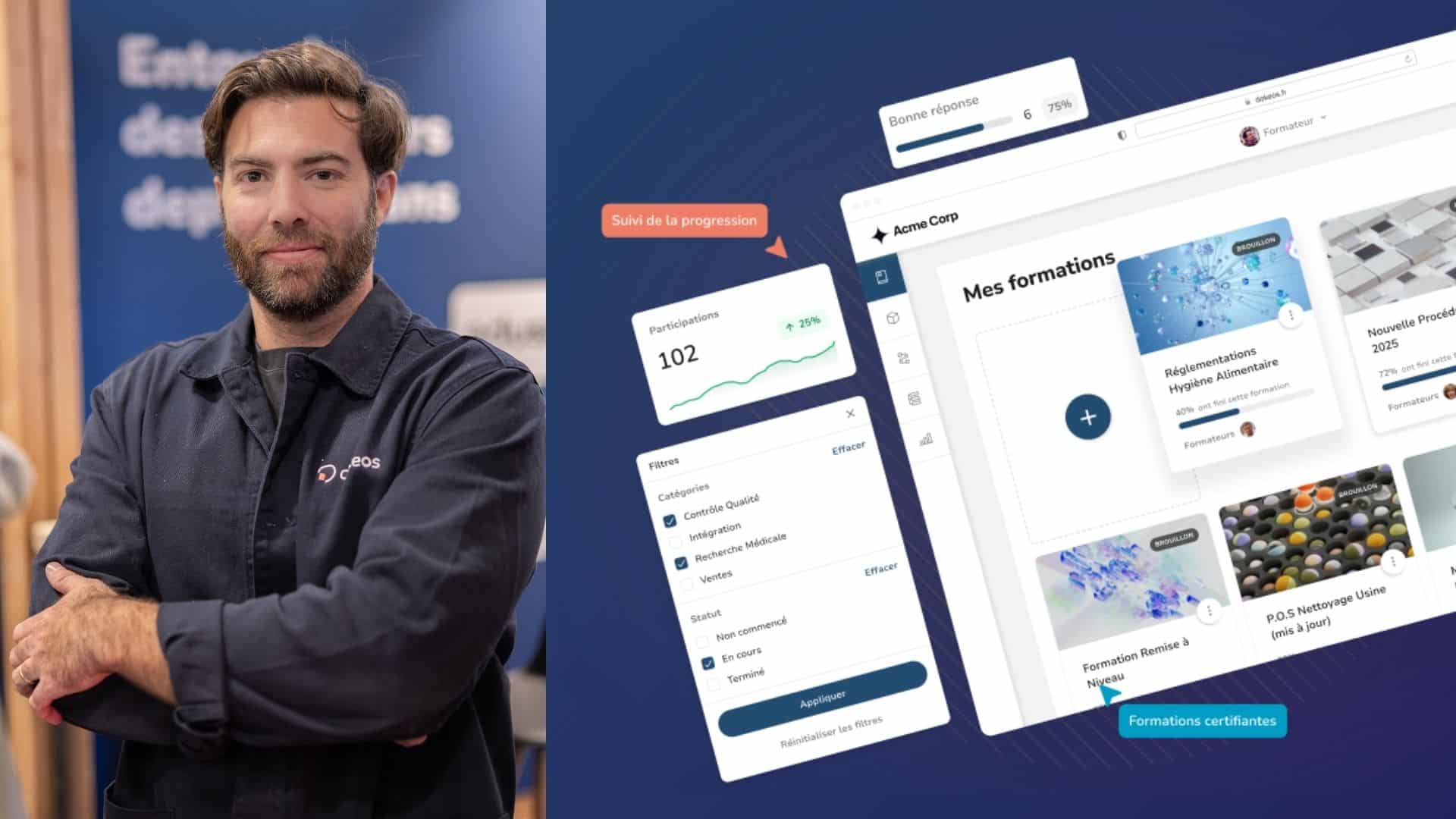
It’s not feasible for pharmaceutical employees to return to a traditional classroom format. This is too time-consuming; it’s also costly for companies and the time away from the work floor can be a major disruptor. More pharmaceutical companies have adopted a remote or hybrid learning environment using Dokeos LMS solutions. Consider these ways that Elearning and Dokeos LMS are reshaping the development of pharma workforce skills.
1. Stay up to date with regulatory requirements
As technology changes, so does official regulations. Globally recognized institutions like the World Health Organization (WHO) and the International Society for Pharmaceutical Engineering (ISPE) establish global industry standards. This is in addition to national regulatory bodies, such as the Food and Drug Administration (FDA) in the U.S. and the Medicines and Healthcare Products Regulatory Agency in the UK.
E-learning provides a one-stop repository where employees can view the latest pharma regulations without having to navigate the official regulatory sites individually. Furthermore, the regulations can be reworded to deviate from the formal and dry language and use simpler wording. Combine this with diverse media, such as videos and infographics for a more engaging learning and understanding.
2. Address a global workforce with Dokeos LMS
More workers in the life science industry are becoming digital natives. 70% of healthcare providers, for example, work almost fully online, according to a 2020 PharmaTimes report. With the remote acquisition, more companies are hiring talent outside their local region, and even hiring globally. No matter where your employees are located, Dokeos LMS can cater to their learning needs. Courses can be translated into different languages without requiring a native speaker to manually rewrite the course. Courses can be accessed 24/7, and webinars can be recorded and viewed at the students’ convenience to accommodate time zone differences.
3. Customize training by region and demographic
Certain facets of the course can also be modified to suit varying demographic needs. This is especially beneficial for pharma companies serving global clientele. For example, approximately half of the world’s senior population resides in the Asia-Pacific zone. Course material for remote employees, affiliates, and joint partners in the region can focus more heavily on lessons related to geriatric and palliative medicine/care.
4. Address security challenges
The pharma sector ranked fifth in industries most susceptible to a cyber-attack, according to Seqrite’s annual 2020 report. The unfortunate reality is that confidential patient data, research data, and intellectual property are prized commodities sold in the black digital market.
Modern LMS platforms like Dokeos LMS reside in cloud networks with the most consistent security updates and patches. The latest encryption and anti-malware scanners are in place to ensure there are no exploitable attack vectors in the network.
5. Accelerated training in times of emergency
COVID-19 was a rude awakening that virus outbreaks can occur rather quickly and with little warning. This proved overwhelming for the medical industry as it raced against time to develop a vaccine, study the changing virus strains, and address overcrowded ER rooms.
With Dokeos LMS, cut down on time-wasting elements like traveling, assembling a classroom, etc. Once the course is live, employees can get started immediately, regardless of location or time difference. Changes can also be rapidly made to learning materials as new research developments come in.
Prioritize workforce learning with Dokeos LMS and GXP Training!
From regulatory compliance to procedural training, Dokeos LMS provides a platform that suits the modern pharmaceutical workforce, whether that be in the office or in the lab. Today’s pharma employees require continuing education to address the ever-changing medical landscape. Our LMS makes education and its administration accessible for employees and HR alike. Sign up for a free trial today!
At GXP Training, we’ve leveraged the insights of elite quality assurance professionals to craft a digital repository of certified regulatory training programs. These courses are intricately tailored to meet the rigorous standards of industries operating under strict regulatory frameworks. Our courses are a testament to their excellence and effectiveness. Our catalog encompasses a wide array of individual certifications, 100% online for your convenience. Additionally, our courses are accredited for CPD (Continuing Professional Development) and CEU (Continuing Education Unit) credits, providing professionals in the pharmaceutical sector with the essential knowledge and credentials required to thrive in this rigorously regulated industry. Explore our catalog today to elevate your career and meet the stringent standards of compliance. Contact us!